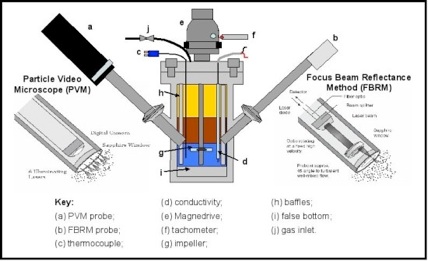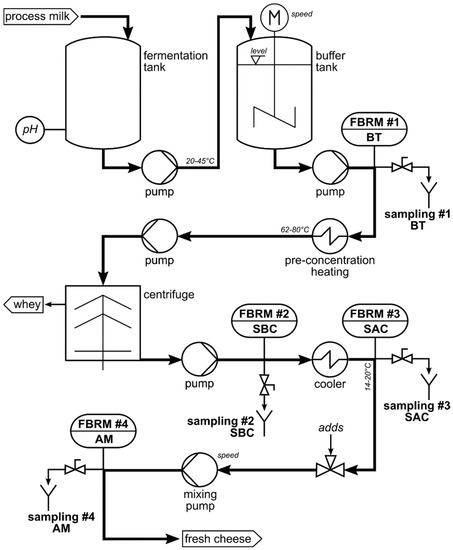
Dairy | Free Full-Text | Inline Particle Size Analysis during Technical-Scale Processing of a Fermented Concentrated Milk Protein-Based Microgel Dispersion: Feasibility as a Process Control

Feasibility of Focused Beam Reflectance Measurement (FBRM) for Analysis of Pharmaceutical Suspensions in Preclinical Development | SpringerLink

Application of a recent FBRM-probe model to quantify preferential crystallization of dl-threonine - ScienceDirect
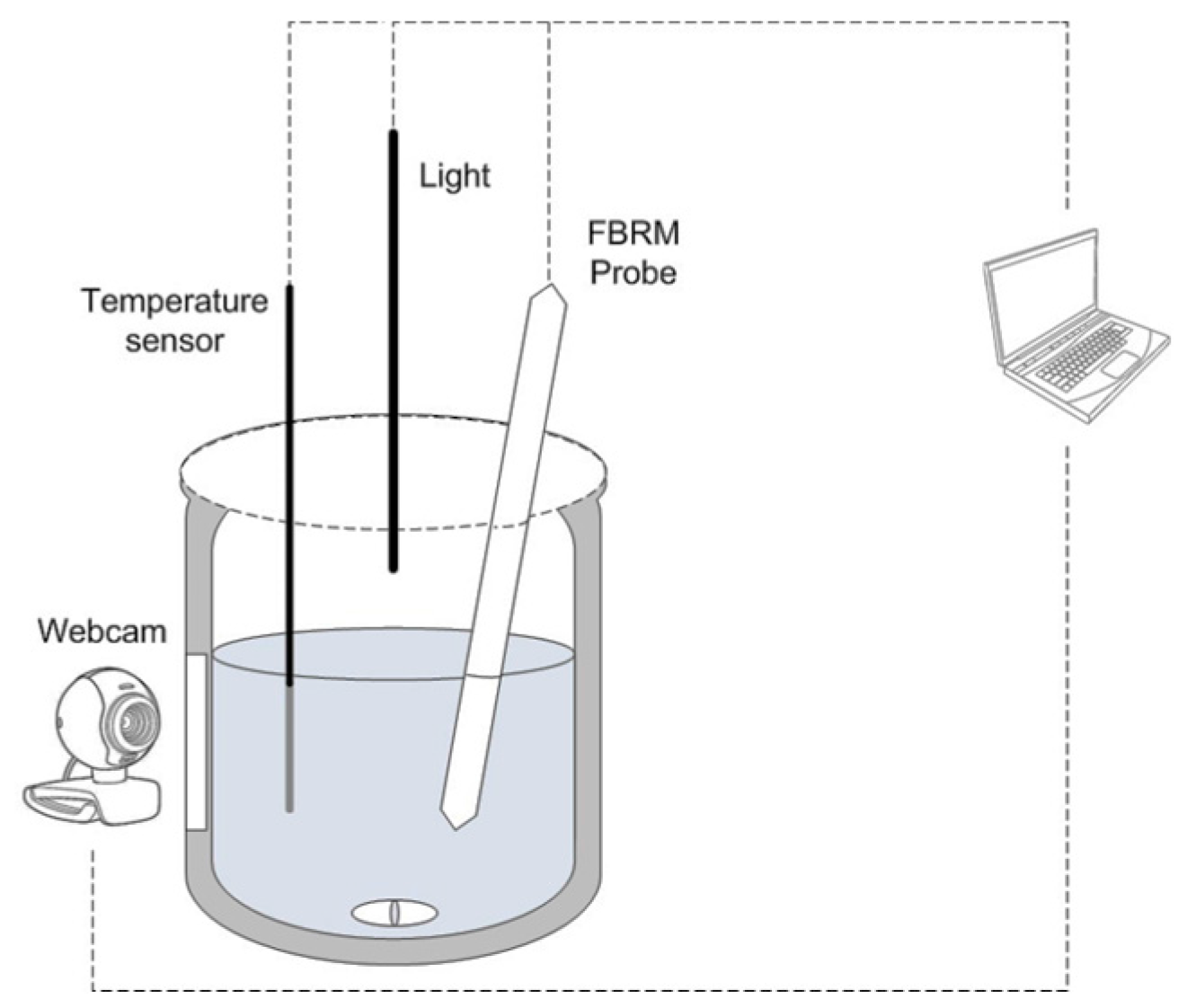
Sensors | Free Full-Text | A Low-Cost System Based on Image Analysis for Monitoring the Crystal Growth Process

Using focused beam reflectance measurement (FBRM) to monitor aggregate structures formed in flocculated clay suspensions - ScienceDirect

Resolution and Sensitivity of Inline Focused Beam Reflectance Measurement During Wet Granulation in Pharmaceutically Relevant Particle Size Ranges - Journal of Pharmaceutical Sciences
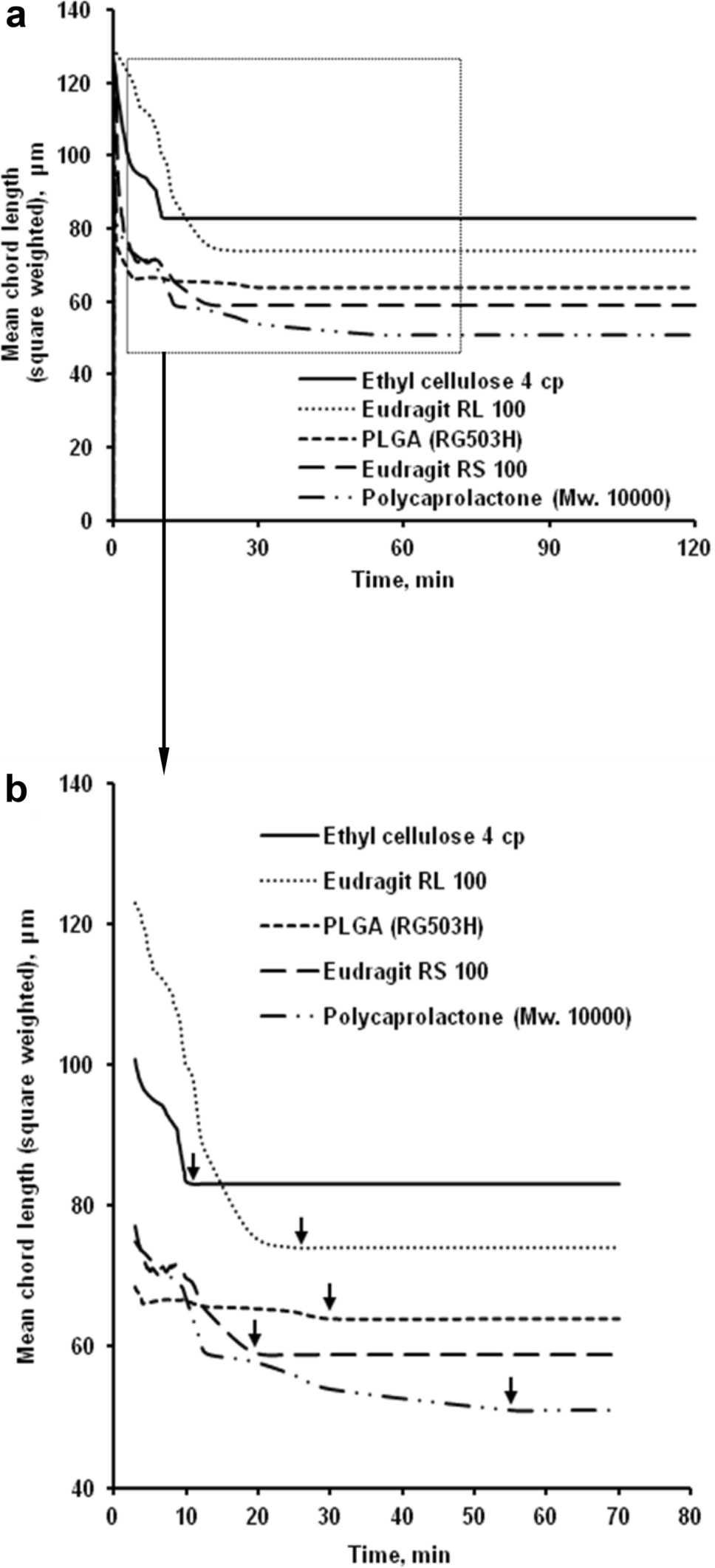
Real-time particle size analysis using focused beam reflectance measurement as a process analytical technology tool for continuous microencapsulation process | Scientific Reports

FBRM principle. (a) FBRM probe, (b) chord length measurement, and (c)... | Download Scientific Diagram

Utilization of In Situ FBRM and PVM Probes to Analyze the Influences of Monopropylene Glycol and Oleic Acid as Novel Additives on the Properties of Boric Acid Crystals | Industrial & Engineering

Working principle of focused beam reflectance measurement (FBRM). (a)... | Download Scientific Diagram

Optimization of a Crystallization by Online FBRM Analysis of Needle-Shaped Crystals | Organic Process Research & Development

a) Measuring principle of focused beam reflectance measurement (FBRM)... | Download Scientific Diagram
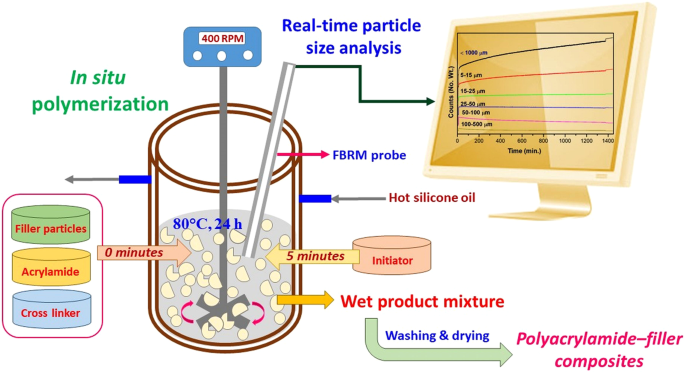
Real-time Particle Size Analysis Using the Focused Beam Reflectance Measurement Probe for In Situ Fabrication of Polyacrylamide–Filler Composite Materials | Scientific Reports

Figure 10 from Feasibility of Focused Beam Reflectance Measurement (FBRM) for Analysis of Pharmaceutical Suspensions in Preclinical Development | Semantic Scholar

Feasibility of Focused Beam Reflectance Measurement (FBRM) for Analysis of Pharmaceutical Suspensions in Preclinical Development | SpringerLink