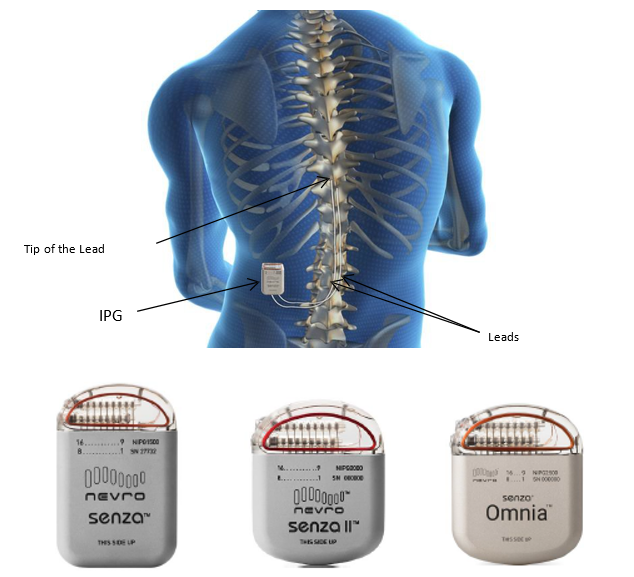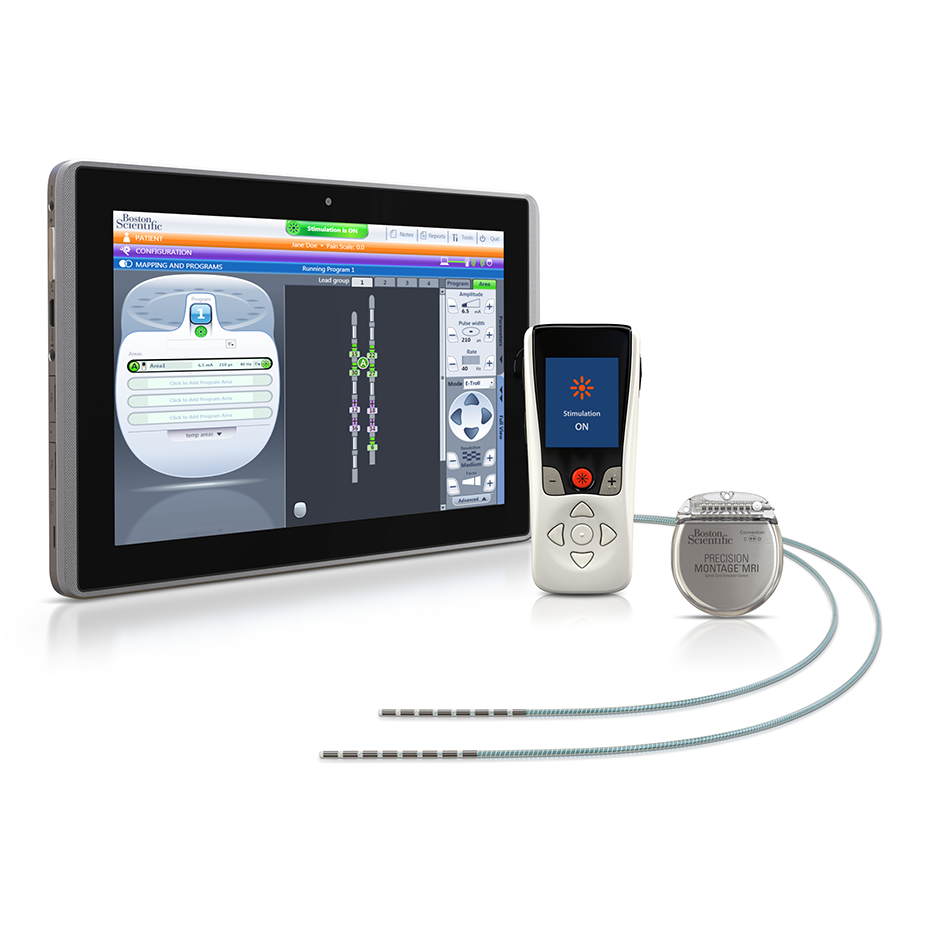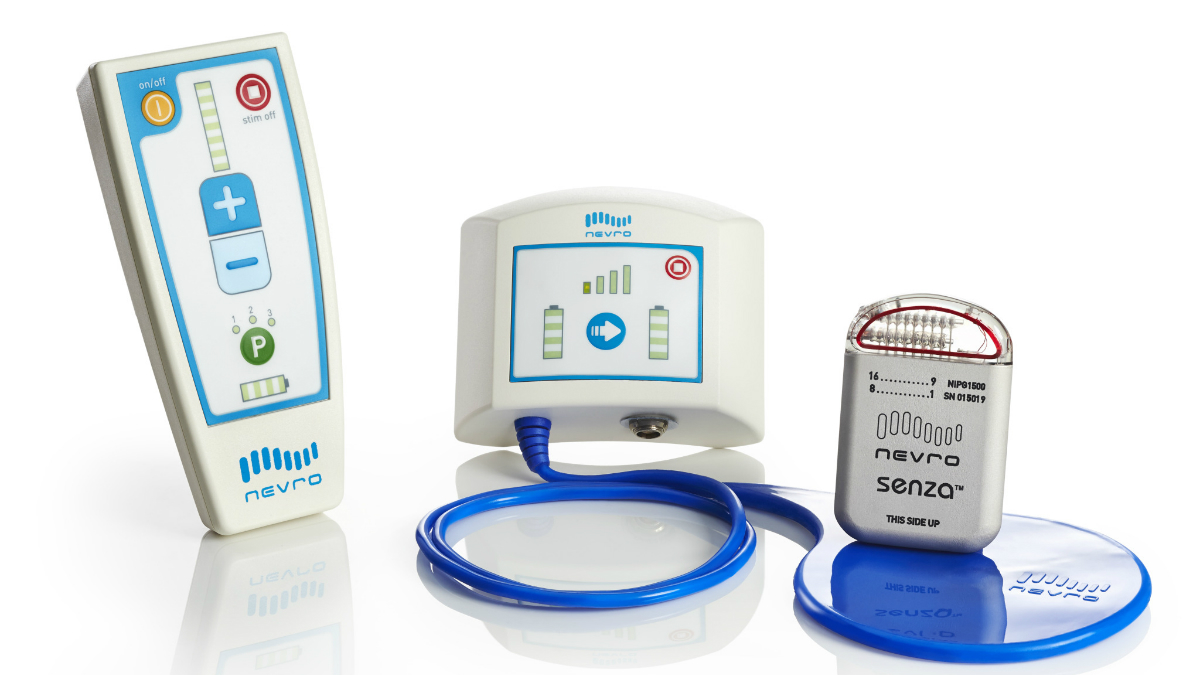
NICE recommends spinal cord stimulation system for chronic back or leg pain | The Chartered Society of Physiotherapy
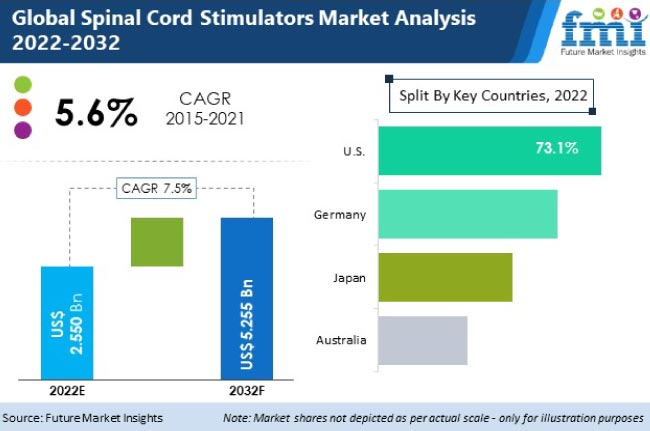
Spinal Cord Stimulators Market is expected to grow at over a CAGR of 7.5% during the forecast period (2022-2032). - PharmiWeb.com

Long-term safety and efficacy of closed-loop spinal cord stimulation to treat chronic back and leg pain (Evoke): a double-blind, randomised, controlled trial - The Lancet Neurology

Nevro Corp. - Nevro Announces U.S. Full Market Launch of Revolutionary HFX iQ™ Spinal Cord Stimulation System to Personalize Treatment of Chronic Pain
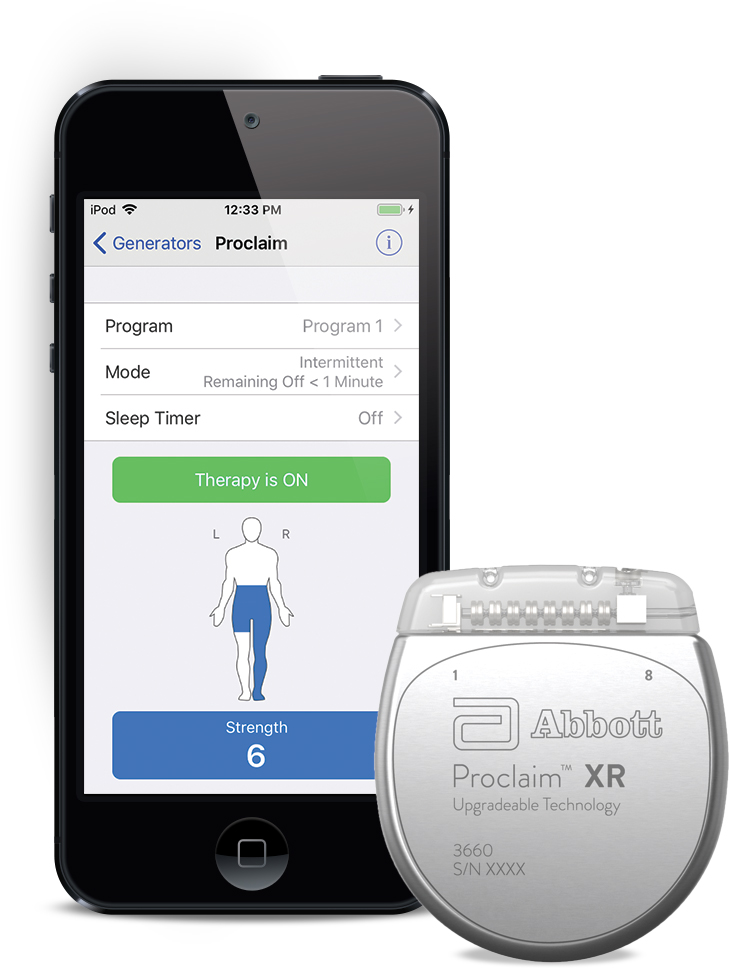
FDA Approves Abbott's "Low Dose," Recharge-Free Spinal Cord Stimulation System with up to Ten Year Battery Life* for People Living with Chronic Pain - Sep 26, 2019